
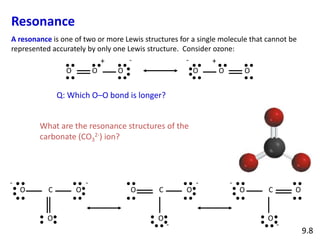
Explain why CO_{3}^{2-} ion cannot be represented by a single Lewis structure. How can it be best represented?

Question 44 Explain why CO 32- ion cannot be represented by a single Lewis structure. How can it be best represented?

All questions go with Model 4 , please label thank you in the Ltwis suettre for PCls and XeFe Model 4: Which Lewis Structure is Better? The two structures below could represent

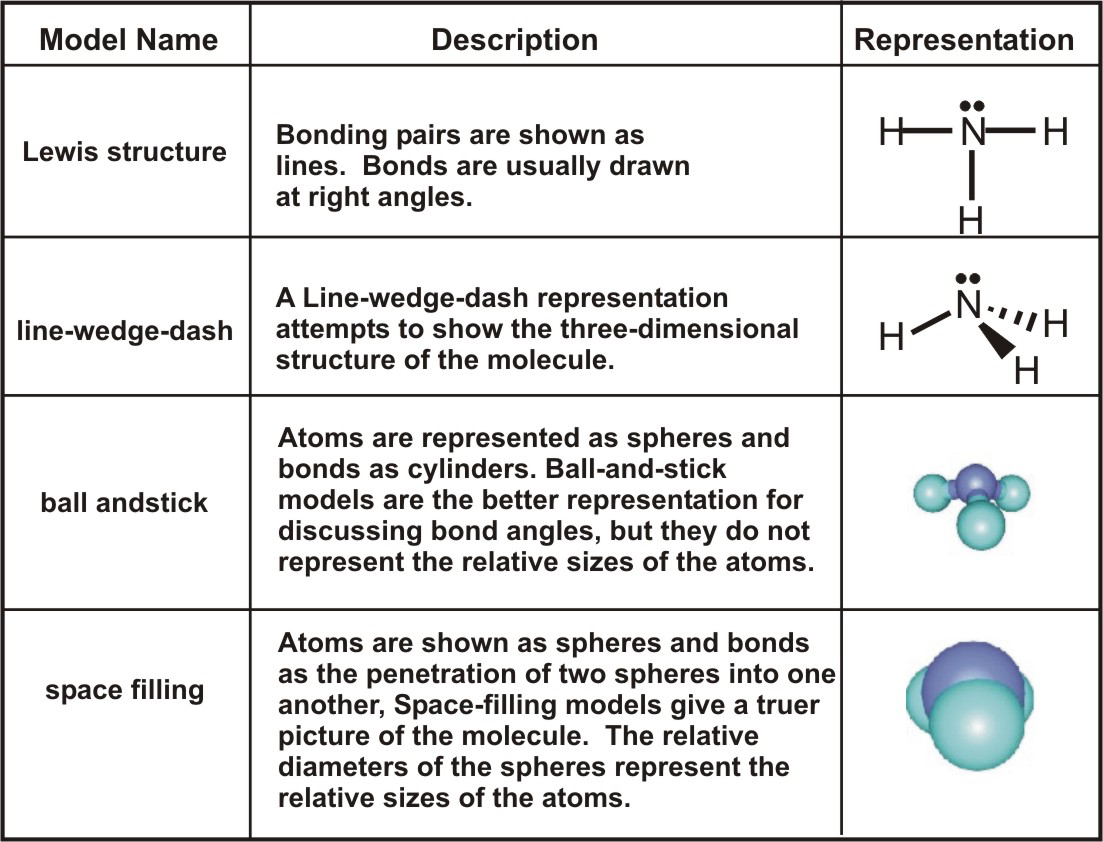
Lewis Dot Structures Lewis Dot structures are used to represent the valence electrons of atoms in covalent molecules Dots are used to represent only the. - ppt download
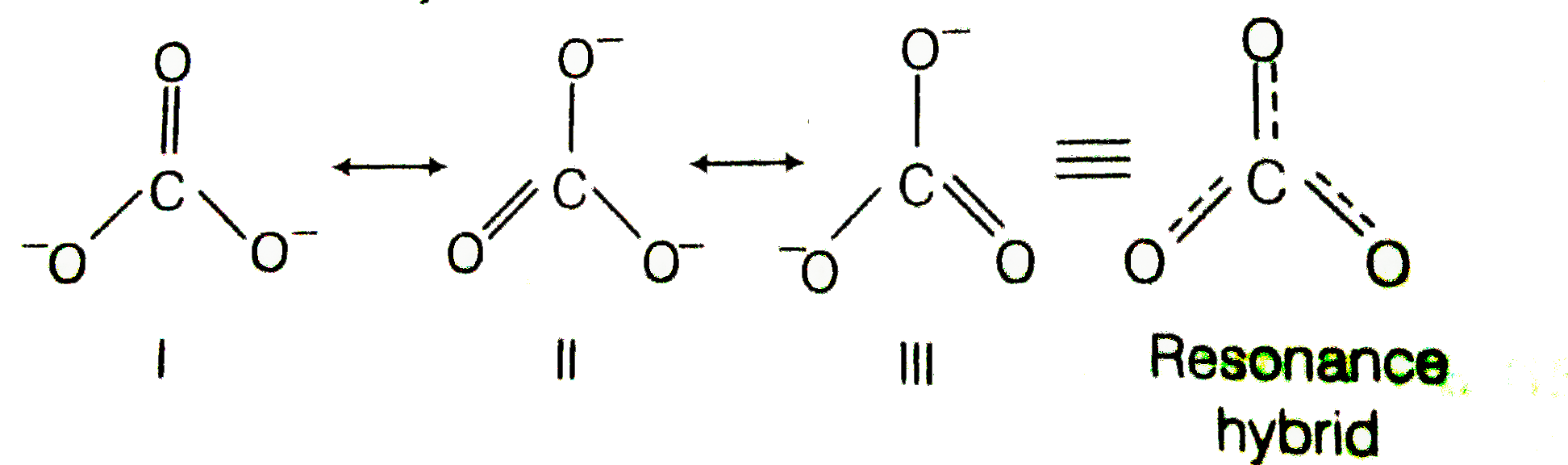
Explain why CO_(3)^(2-) ion cannot be represented by a single Lewis structure. How can it be best represented?





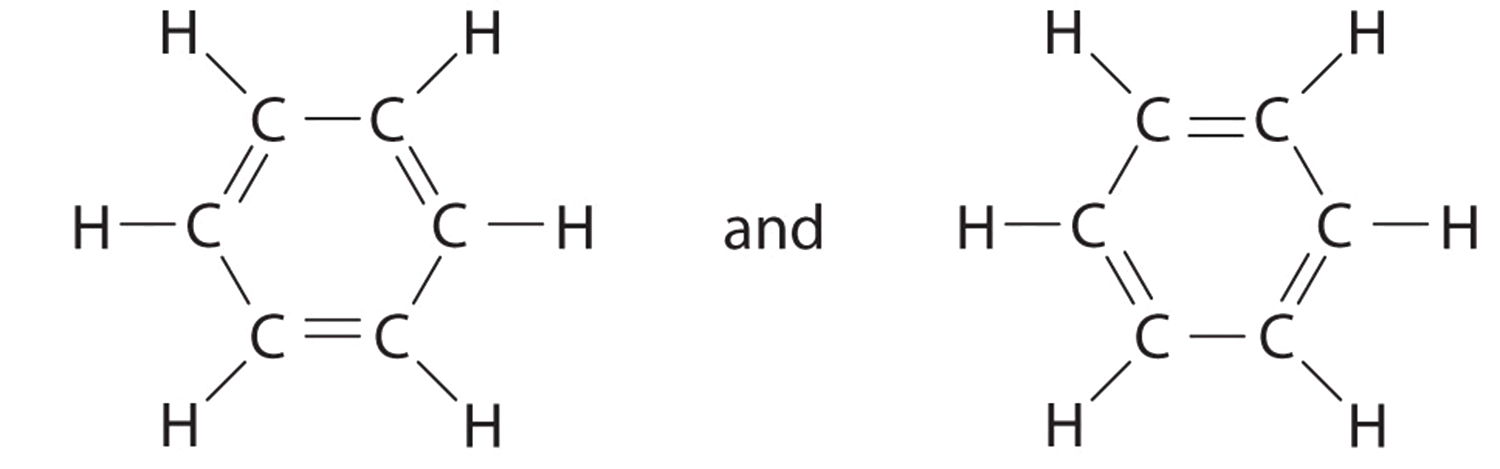



/Lewis-dot-structure-58e5390f3df78c5162b4c3db.jpg)



/ScreenShot2018-11-19at11.40.52PM-5bf3909a46e0fb00510dbd6d.png)



