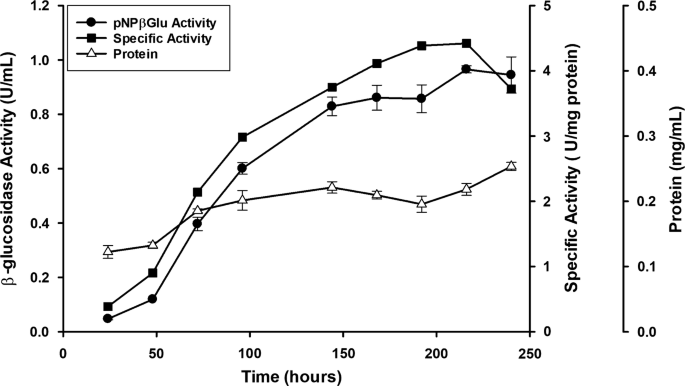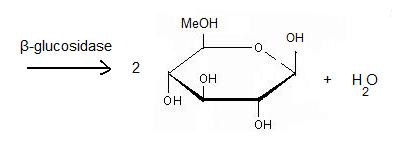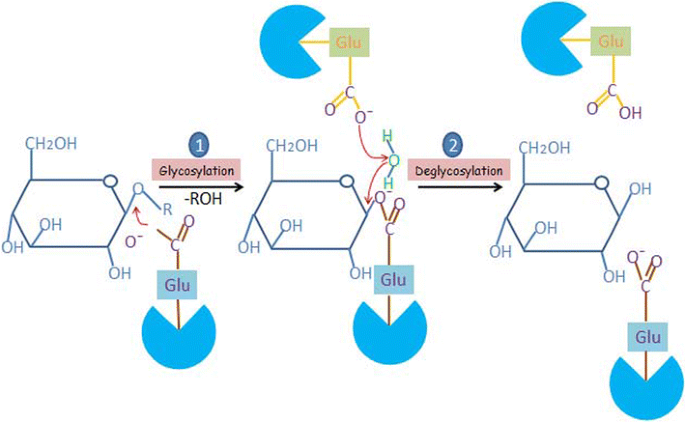
Catalytic properties, functional attributes and industrial applications of β -glucosidases | SpringerLink

Frontiers | Plant Defensive β-Glucosidases Resist Digestion and Sustain Activity in the Gut of a Lepidopteran Herbivore | Plant Science

Characterization of an extracellular β-glucosidase from Dekkera bruxellensis for resveratrol production - ScienceDirect

Overexpression and characterization of a glucose-tolerant β-glucosidase from Thermotoga thermarum DSM 5069T with high catalytic efficiency of ginsenoside Rb1 to Rd - ScienceDirect

Temperature sensitivity of mineral-enzyme interactions on the hydrolysis of cellobiose and indican by β-glucosidase - ScienceDirect
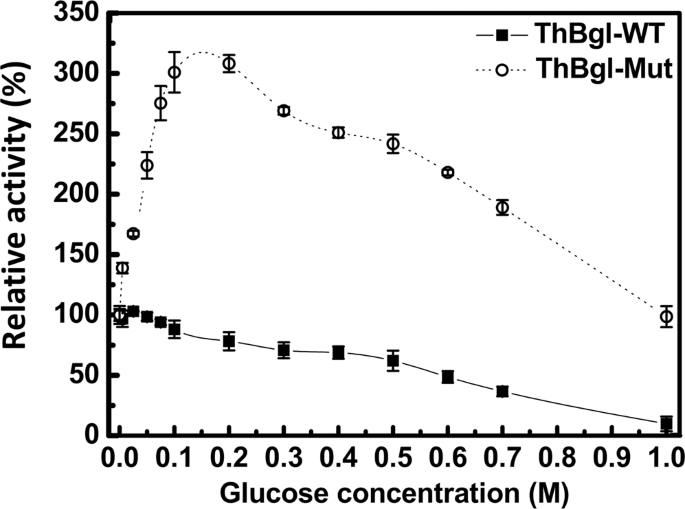
An engineered GH1 β-glucosidase displays enhanced glucose tolerance and increased sugar release from lignocellulosic materials | Scientific Reports

The mechanism of substrate (aglycone) specificity in β-glucosidases is revealed by crystal structures of mutant maize β-glucosidase-DIMBOA, -DIMBOAGlc, and -dhurrin complexes | PNAS
PLOS ONE: Several Genes Encoding Enzymes with the Same Activity Are Necessary for Aerobic Fungal Degradation of Cellulose in Nature
Engineering the GH1 β-glucosidase from Humicola insolens: Insights on the stimulation of activity by glucose and xylose
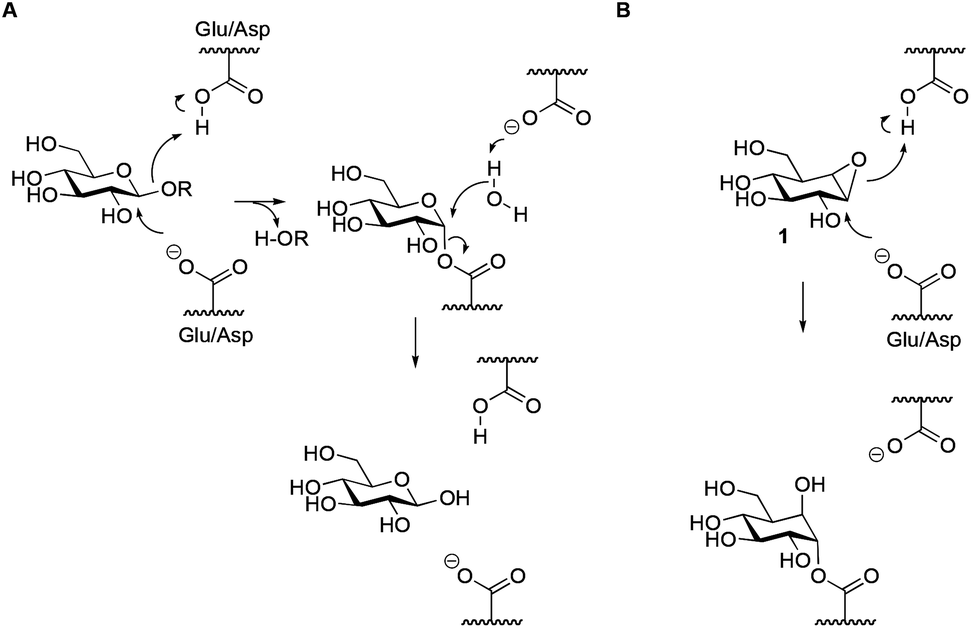
Exploring functional cyclophellitol analogues as human retaining beta- glucosidase inhibitors - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB01611D